Small scale, large impact
4 mins read
How MEMs based systems are finding medical applications. By Graham Pitcher.
Microsystems get called a range of things. But whatever you prefer to the approach – other terms include MEMs – the concept of using semiconductor - like manufacturing techniques to produce ultrasmall devices is getting ever more popular. And one of the first sectors to take up the challenge of applying microsystems is the medical world.
The cost of health care is a continuing worry, whether it's funding the NHS in the UK's case, or arranging suitable insurance in other areas. Being in hospital is a costly business and many countries are working on ways of keeping people out of hospital unless it's absolutely necessary. That needs part of the workload to be moved back into the community, which means to the doctor's surgery.
But there is a problem here. The cost of the equipment used in hospitals, while very capable, is very expensive. The system simply cannot afford to install hospital style equipment in every surgery. So other answers are being pursued and microsystems are a recurring solution.
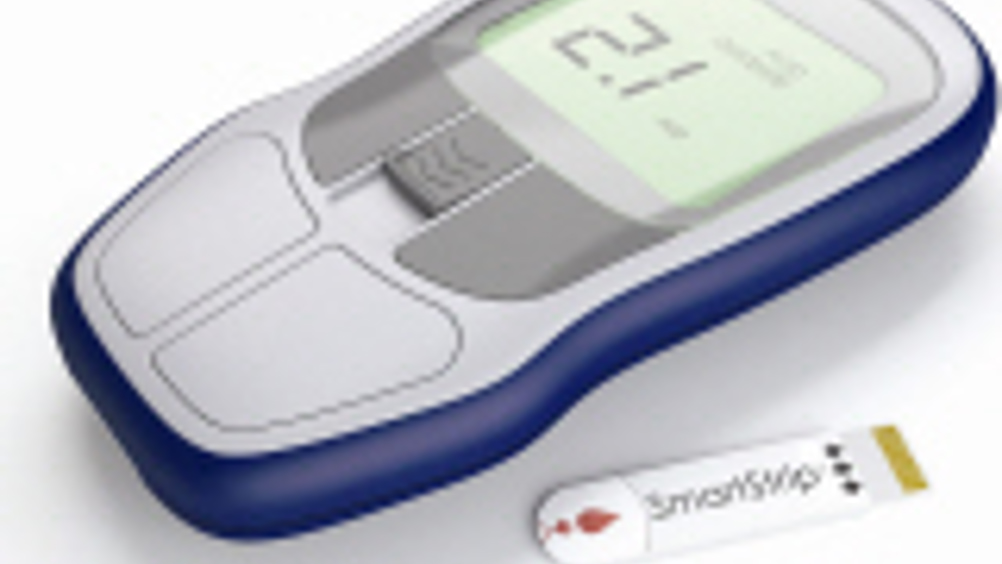
There's a range of tests that can be done in the doctor's surgery, given access to the right equipment. And, with the right answers to hand, the doctor can be more proactive in the patient's treatment. One test which can give insight into a range of conditions is how quickly blood clots and Microvisk is looking to produce a solution using microsystems technology.
Anti coagulant drugs, which delay clot formation, are administered for a number of conditions, including heart problems, so the blood clotting time needs to be monitored regularly in order to maintain a safe dosage level.
According to the company, blood clotting is a crucial bodily function and is currently checked using the Prothrombin Time test.
John Curtis, Microvisk's ceo, explained the process. "Currently, clotting tests are conducted using surrogates – they rely on thrombin generation. But that's not coagulation; it happens only as part of the coagulation process."
The process is based around a seemingly simple cantilever arrangement. "The work was started by Slava Djavok at Cardiff University, then moved to the Rutherford Appleton Laboratory, where development work was undertaken by the central microstructure facility. But Rutherford Appleton lacked the money to commercialise the idea," Curtis continued.
Meanwhile, Curtis – who has long experience in medical diagnostics – was exploring the laboratory's technology portfolio and thought the idea could be commercialised.
The attraction for Curtis is the opportunity to participate in an emerging market. "The coagulation home testing market has the potential to be the same size as that of glucose testing. Although there are fewer people taking warfarin, the tests are currently 10 times more expensive than glucose tests. And, while there are 165 companies offering glucose testers, there are only two others in the coagulation market."
The Microvisk approach works on the blood sample itself. Curtis said: "We measure coagulation as it happens through the change in the blood's viscosity."
He noted that blood has a normal viscosity of around 2cP. "But when it coagulates, viscosity can become 300 to 400cP within 100ms. We can measure this change in real time and that is something no other company can do. Competitors measure biochemistry; we measure physical properties."
Blood properties are determined using a three layer sensor strip. Two of these layers are polyimide, with a labyrinthine piezo sensor layer sandwiched in between. Because the top and bottom layers are of different thickness, their stress is different. "In essence, it's a bimetallic strip," Curtis noted.
The top and bottom layers incorporate precisely positioned heaters which apply a 600µs heat pulse to a 5µl blood sample. "We determine viscosity by measuring the output from the piezo sensor as the strip goes down and then when it springs back up.
Microvisk's sensor is currently being manufactured by Semefab. "We're getting 80 to 85% yield and 100% reliability," Curtis claimed. The cantilevers measure 800 x300µm and the whole package measures 2 x 1mm. "The cantilevers are glued on what is, effectively, a circuit board using a conductive epoxy. We put a capillary layer on top and then a plastic capping layer," said Curtis.
Having already raised £7million, Microvisk is currently in the middle of a new £6m funding round, which Curtis says is 'over subscribed'. While the major thrust for the company is coagulation monitoring, Curtis says the technique can be applied to measuring the viscosity of 'pretty much anything'. "The system has demonstrated linearity from 0 to 12,000cP with a sensitivity of 0.5cP," he claimed, "and it can also measure viscoelasticity and yield stress."
Meanwhile, imec and Holst Centre have developed very sensitive integrated sensing elements for gas detection. The polymer coated microbridges, which are implemented in high density arrays, can detect vapour concentrations at the part per million level using on chip integrated read out techniques. The technology is said to be suitable for the miniaturisation of electronic noses due to its low power consumption and small form factor.
The suspended structures vibrate individually and changes in their modes of vibration provide an indication of vapour absorption in their coatings. Because the microbridges have a very high length to thickness ratio, the gas sensor chip has a high sensitivity to low concentrations of a given vapour.
Mercedes Crego-Calama, e-nose project leader, said the cantilever approach to detection – used in other designs – would not work at small concentrations, simply because the molecules are too light. "Our approach allows MEMs technology to be used," she claimed. "Our approach is more sensitive and does not require an optical system. And using MEMs technology means the device can be smaller."
The sensing elements are double clamped bridges, each coated with 8 to 10 different polymers, which can be applied using ink jet printing. Each polymer is capable of absorbing different molecules. Crego-Calama said: "When a molecule is absorbed, the polymer swells. Two things then happen – the vibration frequency changes and the beam gets stressed."
Elements normally vibrate at frequencies in excess of 100kHa, but this decreases at about 7Hz/ppm of vapour absorbed. By analysing frequency changes across the detector as a whole, the composition of a vapour can be determined.
Crego-Calama said the MEMs system could be used in prediagnostics as well as in monitoring patients' health. In both modes, the sensor detects metabolites – by products of the body's normal processes. "For example," she said, "those who suffer from asthma often over medicate because it is hard to get the dose right. By analysing the patient's breath, a system could determine whether the patient is taking the right dose or needs to increase the amount of medication."
Because the MEMs based system is small – a sensor with 160 bridges can be encapsulated in a 9 x 9mm chip – it is capable of being integrated in a mobile phone.
A more radical application is to make the e-nose small enough to be implanted inside the olfactory tract. Because different diseases produce different metabolites, it may be possible to detect the presence of, say, lung cancer at an early stage.
The approach remains in the development phase for the moment, but working demonstrations are likely to appear within 18months, with commercially available products following later.











